New Healthmasters Product: Hemmla
Healthmasters introduces HEMMLA - "Hormone Stability - Energy - Mood/Brain - Metabolism - Lipid Profile - Anti-oxidant Boost"
- Certain cognitive (brain-related) effects have been reported with statin use. Statin labels will now include information about some patients experiencing memory loss and confusion.
- Increases in blood sugar levels (hyperglycemia) have been reported with statin use. (Including elevations of HbA1c)
- Some medicines may interact with lovastatin, increasing the risk for muscle injury (myopathy/rhabdomyolysis)
Healthmasters HEMMLA Medical Food Greater Results and Clinical Application
- Healthmasters HEMMLA is an omega-3 Phosholipid concentrate.
- Super Critical-Solvent Free Extraction
- Highest Yield of Phospholipid, EPA/DHA and Astaxanthin
- Healthmasters HEMMLA is the First Medical Food Sourced from Krill.
- NDC (National Drug Code) Registered Product
Indications for Use Include: management of Omega-3 Phospholipid deficiency which may manifest into dyslipidemia conditions and/or metabolic syndrome; both of which can include:
- Elevated Triglycerides > 150 mg/dl
Elevated LDL-C> 70 mg/dl
Low HDL-C(men) > 40mg/dl , (women) > 50mg/dl
Insulin Intolerance/Impaired Glucose Tolerance
Healthmasters HEMMLA Clinical Study Experience
HEMMLA precursor, has been administered in a total of 203 subjects in six clinical trials and no serious side effects were observed at a dose up to 6g/day. Clinical studies aspects include:
- Hyperlipidemia
- Omega-3 Index*
- Astaxanthin ORAC
- Arthritis
- Cognitive Disorders
- Premenstrual Syndrome
HEMMLA versus Lovaza (prescription fish oil – ethyl ester) in preclinical
HEMMLA Open-Label Survey
Omega-3 Index: Indicates the amount of EPA & DHA in red blood cells versus total fatty acids.
|
|
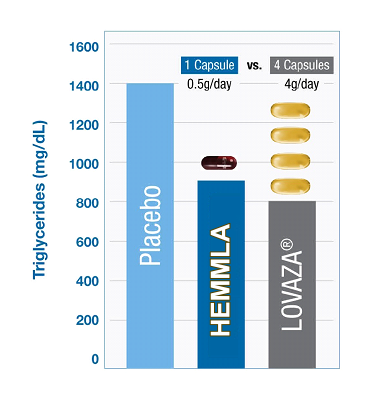
HEMMLA vs. Lovaza (Preclinical Study Experience) HEMMLA has been tested in Zucker Diabetic models of metabolic syndrome for 12 weeks at single daily dosing of 500mg/day (HED; Human Equivalent Dose) and compared to prescribed fish oil. Results: HEMMLA demonstrated equivalent triglyceride reduction with 1 capsule (0.5g/ day) vs. 4 capsules (4g/day) of Lovaza. |
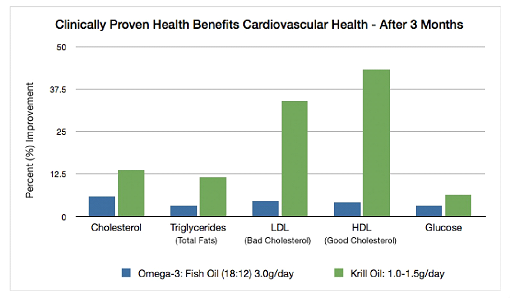
HEMMLA Hyperlipidemia Clinical Study
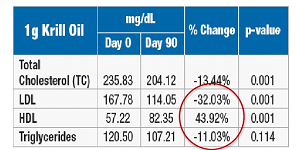 |
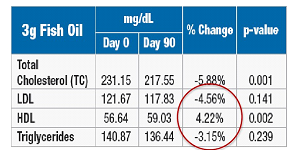
The multi-center, 90-day,
prospective, randomized, double- blind, placebo and active- controlled trial was conducted in 120 patients with mild to moderate hyperlipidemia, comparing placebo to 1 to 3g/day krill oil versus fish oil 3g/day. |
 |
Additional Results:
Blood glucose levels were reduced
by 6.3 percent, from 105 mg/dL to 98 mg/dL (p=0.025), in patients receiving 1 g and 1.5 g krill oil daily. |
HEMMLA Trial Results
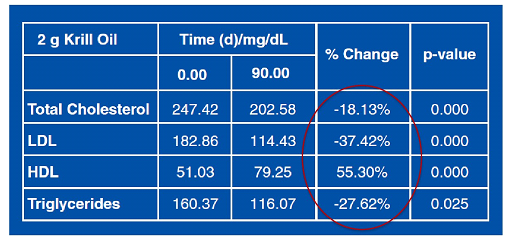
Table 3. Results of Krill Oil (2.0 g/day) on Lipids
HEMMLA Study Summary
Low Dose HEMMLA Longer Term
Table 7.Effect of a Lower Maintenance Dose of Krill Oil on Lipids
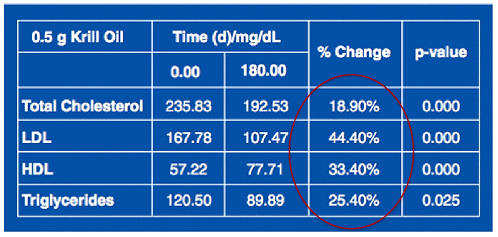
Patients receiving 1 g and 1.5 g (60 people) daily krill oil continued for another 12 weeks with a lower maintenance dose of 0.5 g krill oil daily.
HEMMLA vs. Fish Oil More Absorption & Greater Bioavailability
|
Krill Oil Phosphilids carry Omega-3s (O3s) and are easily recognized, incorporated and utilized by the body’s cells. *Note no cellular energy is needed for this to occur. |
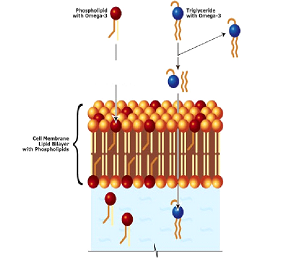 |
Fish Oil Triglycerides carry O3s and are not incorporated into the body’s cell membranes. ATP must be used to break them down before they enter the cell, then they are reassembled and are transported. *Note that TG’s can be stored in adipose tissue as a reserve energy source. |
Astaxanthin in cardiovascular health and disease.
- Experimental investigations in a range of species using a cardiac ischaemia-reperfusion model demonstrated cardiac muscle preservation when astaxanthin is administered either orally or intravenously prior to the induction of ischemia.
- Astaxanthin lowered oxidative stress in overweight and obese subjects and in smokers. It blocked oxidative DNA damage, lowered C-reactive protein (CRP) and other inflammation biomarkers, and boosted immunity in the tuberculin skin test.
- Astaxanthin has a unique chemical structure allowing it to span the entire width of the cell membrane.
- By doing so, it provides powerful antioxidant protection on both sides of the cell membrane as well as the interior of the cell membrane.
- Other antioxidants cannot do this; they can only exert protection in specific locations throughout the cells.
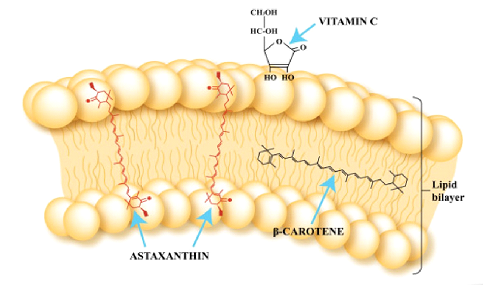
Astaxanthin
- Antioxidant: 65 times more powerful than vitamin C; 54 times stronger than beta carotene; 14 times more potent than vitamin E
- Anti Inflammatory
- Insulin Sensitizer (as is PPL)
Case Study - Statin & Healthmasters HEMMLA
60-year-old female, referred for an abnormal lipid profile. On presentation, lipid profile showed in mg/dL:
TC/HDL = 6.56Trig = 395LDL = 160
Simvastatin 40mg with a lipid profile (mg/dL) 6 weeks later showing:
TC/HDL = 6.32 (-4%)Trig = 215 (-46%)LDL = 90 (-44%)
HEMMLA 1gm was started. Six weeks later a lipid profile showed in mg/dL:
TC/HDL = 3.35 (-47%)Trig = 178 (-16%)LDL = 58 (-35%)
Disclaimer: Case studies represent results of a single participant. Consumers should weigh these results with other scientific data and should not expect the same results as those found in case studies alone.
Healthmasters HEMMLA Precursor PMS Study
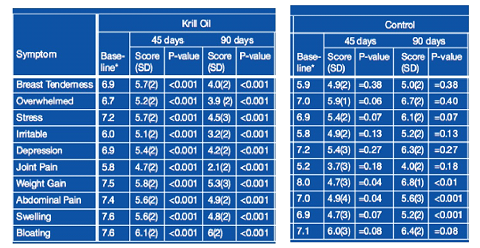
Healthmasters HEMMLA Precusor Krill Oil can significantly reduce dysmenorrhea and the emotional symptoms of premenstrual syndrome and is shown to be significantly more effective for the complete management of premenstrual symptoms compared to omega-3 fish oil.
|
Clinical Study Experience HEMMLA’s main component (NHP80006416 / GRN242 †) has been administered in a total of 203 subjects in 6 clinical trials and no serious side effects were observed at a dose up to 6g/ day |
Omega 3 Phospholipid Source for HEMMLA

Antarctic Krill (Euphausia superba)
FDA Standards for a Medical Food
- Shrimp-like crustacean (bottom of food chain)
- Most Abundant marine biomass (> 500 million tons)
- Consumed by various marine-life – whales, fish, etc.
- Global capture = 5.5% allowed quota - human health benefits < 1%
- Rich source of Omega-3 Phospholipids
- Certified by Friends of the Sea (FOS), an international organization that verifies sustainable origin of marine products
-
- Defined by the FDA (in section 5(b) of the Orphan Drug Act (21 U.S.C. 360ee (b) (3))) as "a food which is:
- formulated to be consumed or administered internally under the supervision of a physician
- intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation
- designated Generally Regarded As Safe (GRAS).
-